The guest post below was written by Jacob Bruggemann, a graduate student based at the Scripps Research Institute, who helped lead the biomedical Zooniverse project, Microscopy Masters.
Read on to find out about the findings of this project, made possible by the efforts of our fantastic citizen scientist volunteers. If you would like to learn more, you can access a preprint publication about this work here.
With thanks to Zooniverse Volunteer Becky Kennard for editing this piece.
– Helen
Microscopy Masters draws to a close
Microscopy Masters, the cryo-electron microscopy (cryo-EM) project building complex 3D models of proteins, is approaching its initial conclusion. Over the two years the project has been running, we’ve collected over 17,000 classifications and built a dataset of 209,696 unique protein particles. The primary dataset used in this project was the 26S proteasome lid complex generated by the Lander Lab at the Scripps Research Institute. We have also annotated other, smaller datasets.
The proteasome is a large multi-protein complex responsible for breaking down unwanted proteins into reusable parts, kind of like a large recycling center for the cell. Studying its structure could reveal the mechanisms behind how the proteasome lid only opens for proteins marked for recycling, and give insights into problems caused by the lid malfunctioning.
This project is centered on an important tenet in biology, ‘form follows function.’ On the molecular level of biology, what this means is that the shapes of large biological molecules, such as proteins and nucleic acids, are evolved to perform specific functions. By studying and understanding the structure of biological complexes, researchers can better understand how all the little moving parts of life interact, which will allow them to better combat diseases and disorders.
Scientists are often too busy in the lab to come up with catchy names, meaning that the techniques they invent are usually given pretty self-explanatory titles. In the case of cryo-EM, everything you need to know is in the name.
Imagine some scientists are interested in studying a protein, say our subject, the 26S proteasome lid. Cells containing large quantities of the protein are lysed (a scientific way to say ‘popped like balloons’) and the contents of the cells are put into a solution. That solution is purified so that it only contains the protein the scientists are interested in. The purified solution is then flash-frozen in extremely thin ice (cryo) and put under an electron microscope (EM) to obtain images of the proteins. These images are then put through sophisticated reconstruction software to obtain a detailed 3D model of the protein. This technique is so powerful, scientists can identify individual atoms in the protein complex, giving them deep insights into how it interacts with its environment.
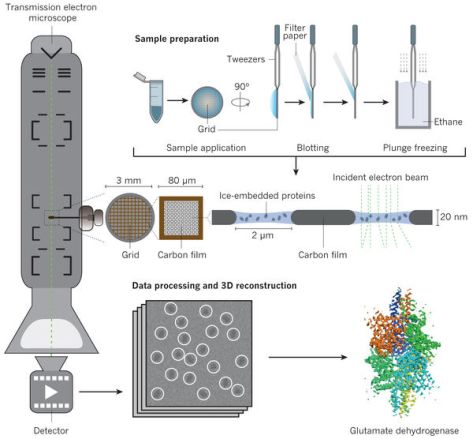
Figure 1 A schematic of how cryo-EM is done. Taken from dx.doi.org/10.1038/nature19948.
Of course, there have been years of work involved in the cryo-EM that I just explained in four sentences (and some very, very expensive microscopes!). A particularly time-consuming task for cryo-microscopists is picking the individual proteins from the microscopy images (micrographs), called ‘particle picking.’
Scientists used to do this by hand, but since they often have thousands of these images to process, this can take weeks of work. So, they usually rely on software to extract the protein images. But because some proteins are so complex, it can be difficult for software to identify them in the noisy micrographs. For this reason, we decided to train citizen scientists to pick the particles from our proteasome lid data and see if it could be used to build a detailed molecular model.

Figure 2 On the left is a blank micrograph, on the right is a micrograph that has had the proteins manually picked.
Using the data from our volunteers, we made a full 3D reconstruction of the proteasome lid. We compared the model to one made with an automatic particle picker, both of which are shown below. Although they look very similar, what matters for microscopists is the ‘resolution’ of the reconstructions. What resolution means in this context is how consistent the models are when used several times.
In this case each dataset was divided into two random halves and made into two separate models, which were then compared to determine a resolution. Even though the resolution for the computer-made model is lower (better) in this case, this is partly due to the fact that the computer-picked dataset had so many more particles. For this reason, we also did a reconstruction using a subset of the computer-picked data with the same amount of particles as the crowdsourced dataset. This brought the resolution to 4.036 Å, closer to the crowdsourced dataset but still lower.
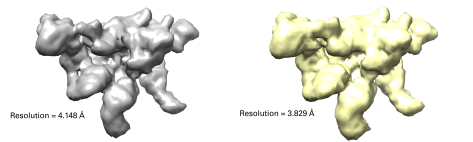
Figure 3 The final reconstructions of the crowdsourced and computational datasets. The resolutions for each are listed, the lower the better.
Even with the higher resolution, we believe this is a fantastic example of the power of citizen science. We built an entirely hand-picked dataset from people who had little to no experience with cryo-EM. This dataset allowed us to build a detailed, 3D model of a complex protein that had a similar resolution to the one built by a team of trained scientists with state-of-the-art software.
This was the first time we have run a project of this nature, and we believe that with tweaking and better feedback systems (which were only implemented by the fantastic Zooniverse team late into our project’s run) we can process data better and faster than we did in our first run.
As a side experiment to try and figure out how to better engage users, some of our project’s participants might remember being sent newsletters about ‘sprint’ datasets, which were small datasets of 15-20 images of other proteins. The use for these was not to build an entire particle dataset, but to provide data for the researchers to feed their automatic particle picking software. We found that giving the images different color schemes than the traditional black-and-white was a nice way to ‘spice’ the micrographs up for users, and we were able to provide researchers with usable data in a matter of days that they could use to start their data processing.
Although we currently have put the Microscopy Masters project on hold, we are excited with the results and in the process of submitting a publication of our initial results. I would like to thank everybody involved with the Zooniverse for building a fantastic platform to try our project. In particular, I would like to thank the Zooniverse team for answering my questions and helping me get Microscopy Masters up and working.
And lastly, thank you to all the hard-working participants in Microscopy Masters and everybody who participates in this great website!
Find out more:
You can check more of the great work done here at the Su lab on our website.
The Lander Lab is consistently pushing the work being done in cryo-EM, go see the work they are doing at their homepage.
Our publication can be previewed as a preprint on bioRxiv.
